Future Trends and Bioburden Testing Market Forecast 2031
The healthcare and life sciences sectors are undergoing a massive shift toward automation and real-time data integration. As sterilization standards become more complex, the Bioburden Testing Market Forecast indicates a period of sustained expansion. Manufacturers are moving beyond simple compliance, treating microbial enumeration as a strategic asset to prevent batch failures and ensure patient safety.
Download Sample Report - https://www.theinsightpartners.com/sample/TIPRE00003000
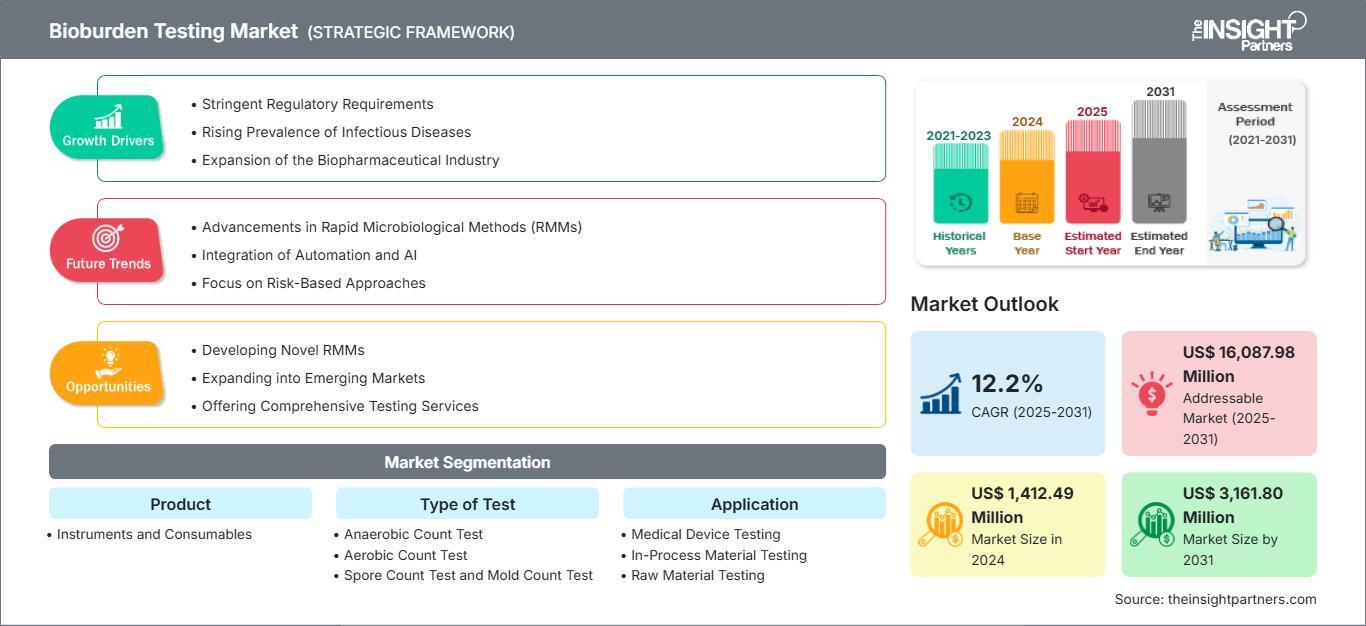
The Bioburden Testing Market Forecast highlights that the market size is expected to reach US$ 3,162 million by 2031. The market is anticipated to register a CAGR of 12.2% during 2025–2031. This growth is driven by a global push for higher manufacturing standards and the rapid adoption of biologics.
Key Market Report Drivers
According to the latest Bioburden Testing Market Forecast, several high-impact factors are accelerating the demand for microbial testing solutions:
1. Heightened Regulatory Scrutiny on Sterility Assurance Regulatory agencies like the FDA and EMA are increasingly focusing on "quality by design." This means manufacturers must monitor microbial loads at multiple stages of production, not just at the end. The Bioburden Testing Market Forecast suggests that the trend of rigorous "in-process" testing will remain a primary driver, as companies strive to avoid the high costs associated with product recalls and regulatory warning letters.
2. Rapid Growth of the Biologics and Cell Therapy Pipelines Biopharmaceutical production involves highly sensitive living cells that are prone to contamination. Traditional sterilization methods can sometimes damage these fragile products, making precise bioburden monitoring essential. The Bioburden Testing Market Forecast identifies the rise in personalized medicine and monoclonal antibodies as a major catalyst for the increased sale of specialized consumables and automated testing platforms.
3. Integration of Rapid Microbiological Methods (RMMs) Time is a critical factor in modern manufacturing. The Bioburden Testing Market Forecast points toward a significant shift from traditional 14-day culture methods to Rapid Microbiological Methods (RMMs). Technologies like ATP bioluminescence and PCR-based assays provide results in a fraction of the time, allowing for faster product release and improved supply chain efficiency.
Leading Players in the Bioburden Testing Space
The competitive environment is evolving as top players invest in AI-driven colony counters and high-throughput screening tools. The following companies are central to the Bioburden Testing Market Forecast through 2031:
- Thermo Fisher Scientific Inc.
- Merck KGaA
- bioMérieux SA
- BD (Becton, Dickinson and Company)
- QIAGEN
- SGS SA
- Pacific BioLabs
- Nelson Laboratories, LLC
- Charles River Laboratories
- STERIS
Regional and Segment Insights
North America remains the largest revenue contributor, supported by a dense concentration of biotech hubs and advanced laboratory infrastructures. However, the Bioburden Testing Market Forecast predicts that Asia Pacific will experience the fastest growth. This is due to the expansion of pharmaceutical manufacturing in India and China, alongside increasing investments in local GMP (Good Manufacturing Practice) compliant facilities.
In terms of segments, the Bioburden Testing Market Forecast shows that consumables—such as culture media and reagents hold a dominant market share. This is attributed to the recurring nature of these purchases. Meanwhile, the instruments segment is gaining ground as labs transition to automated microbial identification systems to reduce human error and increase testing capacity.
Conclusion
The Bioburden Testing Market Forecast for 2031 paints a picture of an industry at the intersection of safety and innovation. With a projected value of US$ 3,162 million and a CAGR of 12.2%, the market is set to play a pivotal role in the future of sterile manufacturing. Companies that embrace rapid testing technologies and automated workflows will be best positioned to navigate the increasingly complex regulatory landscape of the next decade.
Related Report :
· Pharmaceutical Quality Control Market Overview, Growth, Trends, Research Report (2021-2031)
Contact Information -
Email: sales@theinsightpartners.com
Phone: +1-646-491-9876
Also Available in :
Korean German Japanese French Chinese Italian Spanish
- Art
- Causes
- Crafts
- Dance
- Drinks
- Film
- Fitness
- Food
- Oyunlar
- Gardening
- Health
- Home
- Literature
- Music
- Networking
- Other
- Party
- Religion
- Shopping
- Sports
- Theater
- Wellness